Plaintiff wins Bellwether Xarelto Case in Philadelphia Court
A Philadelphia jury awarded Lynn Hartman $27.8 million after finding drug manufacturers Johnson & Johnson and Bayer failed to warn about the dangers of their blood thinning drug Xarelto. The verdict includes $26 million in punitive damage, which juries award to plaintiffs when they believe the defendant acted egregiously. This bellwether case tried in Pennsylvania […]
Shellers honored as National Liberty Museum’s “2017 Heroes of Liberty”

The National Liberty Museum honored Stephen and Sandra Sheller as “2017 Heroes of Liberty” at the 18th Annual Glass Auction & Gala held on October 7, 2017 at the Sheraton Philadelphia Downtown Hotel. The Shellers were recognized for their support of the Museum and the community through their nonprofit organizations The Sheller Family Foundation and […]
SCOTUS Ruling Increases Risperdal Suits in PA

Risperdal litigation in Philadelphia courts have increased significantly this year and the trend is likely to continue. Mass torts against Janssen, a Johnson & Johnson subsidiary, over it’s antipsychotic drug Risperdal has nearly tripled in Pennsylvania. Plaintiff attorneys say the biggest cause for the recent influx is the expiration of a tolling agreement between Janssen […]
Checkbook Science

In 1984 a federal jury in San Francisco found Dow Corning’s breast implants were “defectively designed and manufactured.” Maria Stern was awarded $1.7 M after internal corporate memos and studies show that Dow had failed to warn the public of the health risks associated with breast implants. Evidence from the case was sealed by the […]
The Chamber: ‘One More Bullet in J&J’s Gun’

In ‘For Plaintiffs Bar, Taking on J&J Means Battling a Shadow Foe,’ Law.com’s Amanda Bronstad reports on the “intimate relationship” between Johnson & Johnson (J&J) and the U.S. Chamber of Commerce (the Chamber). The U.S. Chamber of Commerce is not a government entity, but a powerful group of lobbyists, the largest in the nation. Plaintiff’s lawyers point out […]
Breast Implant Safety Controversy Fueled by Junk Science
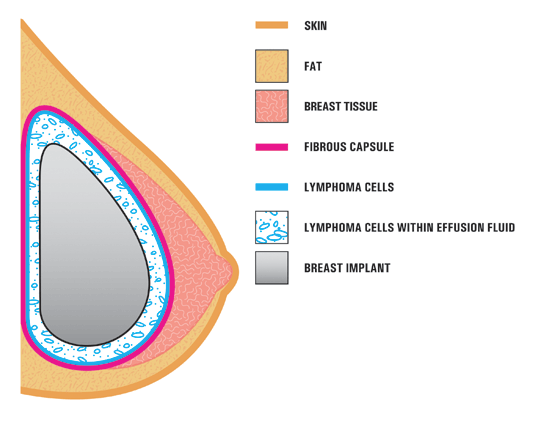
A new type of lymphoma called breast implant-associated anapestic large-cell lymphoma has medical authorities finally agreeing that the prosthetics can cause disease. The first recognized case was in 1997, however a warning from the U.S. Food and Drug Administration was not released until 2011. Six years later, experts say awareness among doctors is low and most […]
Sheller hits #1 Top 50 Verdicts in Pennsylvania for 2016

Sheller PC’s $70 Million Risperdal win hits #1 on the Top 50 Verdicts in Pennsylvania in 2016. In the case A.Y., Pro Ami v. Janssen Pharmaceuticals, Inc., a jury found for an autistic Tennessee boy who grew female-like breasts after taking the antipsychotic drug Rispderal. The unprecedented $70 Million verdict in damages for physical disfigurement and emotional […]
Shellers Honored 2017 “Heroes of Liberty”, National Liberty Museum

On October 7, 2017, Stephen and Sandra Sheller will be honored by the National Liberty Museum as their 2017 “Heroes of Liberty” at the Glass Auction & Gala. This event will support the NLM’s education and outreach programs, something the Sheller’s have both personally, and through their non-profit Sheller Family Foundation, has long supported. The […]
Bard Blood Clot Filter FDA application: forged signature?
Here’s an update on our earlier post about IVC heart filter injuries. Sheller, P.C. medical device attorneys continue to investigate injuries and deaths related to the IVC Blood Filters and are available to discuss legal options with those affected by the devices. Call 800-883-2299 or fill out the inquiry form. NBC News conducted an in-depth […]
Essure Birth Control: Update
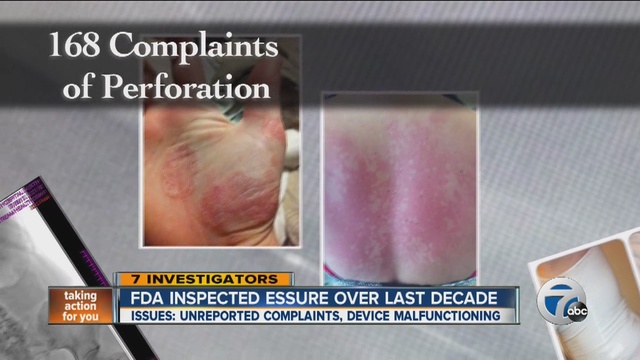
Long term data about the Essure contraceptive device suggest that it may pose risks of serious complications. Essure is a birth control device that consists of two metal coils placed inside the fallopian tubes to block conception. Thousands of women across the country have reported serious side effects associated with the device, including severe and/or chronic pain, […]





